This article was first published in Beitr. Biol. Pflanzen 72, (2000), 1-13.
Deutscher Text in Vorbereitung
Food Composition of Aquatic Bladderworts (Utricularia, Lentibulariaceae) in Various Habitats
By Nikolai Mette¹, Norbert Wilbert¹, Wilhelm Barthlott²
¹Institut für Zoologie der Universität Bonn,
²Botanisches Institut und Botanischer Garten der Universität Bonn
Keywords: Utricularia, prey capture, carnivorous plants
Summary
Till now, only a few authors studied the prey animals of the carnivorous plant genus Utricularia L. In this work, 144 bladders of 3 species (U. australis, U. vulgaris, and U. macrorhiza) from 5 biotopes in Middle Europe (West Germany) were investigated. The prey was determined and counted. To find out the origin of the prey, we also examined the sessile and vagile Aufwuchs colonising the surface of the plants (Periphyton) as well as the plankton out of the habitats.
Most of the bladders were filled with prey. According to the habitats, the prey composition differs with regard to species and quantity. Aquatic Utricularia-species prey on a large variety of food, which remarkably originates from the Aufwuchs of the plants: Cyclopoid copepodids, cladocerans, ostracods, and rotifers, and also insect larvae living in the Aufwuchs and small autotrophic organisms.
Typical plankton organisms from the pelagic zone are not trapped.
1. Introduction
In contrast to the numerous publications on trapping mechanism and ecophysiology of Utricularia (references to find e. g. in Juniper et al. 1989 and Taylor 1989), only a few authors studied the potential or factual prey animals, as did Darwin (1875). The possibly most extensive examination was realised by Garbini (1899), who opened bladders of U. australis [Syn. neglecta] and determined the animalcules inside them. With the same aim, Andrikovics et al. (1988) worked with U. vulgaris from Lake Fertö. Botta (1976) opened traps of three Argentinean species.
The objective of this investigation was to specify the influence of habitat upon the food spectrum of different aquatic Utricularia species.
It was of special interest to compare the prey with the plankton and the Aufwuchs. Aufwuchs is called the organism community settling on animated or inanimate substrate; it consists of sessile organisms as well as vagile animals either feeding on the sessile forms or living between them (Wilbert, 1969). In the following, the term Aufwuchs is reduced to those organisms using the surface of the plants as substrate (Periphyton).
2. Materials and methods
2.1. Plant material studied
Utricularia vulgaris L. — fixated material from two ponds in the nature reserve Gagelbestand, Stallberger Teiche, Lohmar (near Bonn): “Pond 1” and “Pond 2”. The examined plants grew in the reed belts between Phragmites communis.
Utricularia australis R.Br. [Syns. U. neglecta Lehm., U. major Schmidel] — living and fixated plants from two waters inside the Botanical garden of the University of Bonn: an oligotrophe brown water pond (pH 5,5) of an artificial fen biotope (lower fen) and a 5 m² segment of an aquatic plant pond (concrete) with a dense Chara globularis population (pH 8,5).
Utricularia macrorhiza LeConte — living and fixated specimen out of an artificial pond (Pond “Bienenkunde”) from the ground of the Institut für Landwirtschaftliche Zoologie und Bienenkunde in the Melbtal (Bonn), which is grown with Nymphea alba and Potamogeton natans (pH 7,5).
2.2. Faunistic inventories of the habitat
Plankton
10 l water each were drawn with a pail from the fen biotope and from the Pond “Bienenkunde” at the same level at which the bladderworts were growing. The water was filtered directly through a plankton net no. 28. The filtrate was fixed immediately in 4 % formalin.
The registration took place by careful examination of the whole filtrate with the microscope (magnification 4×12.5).
Aufwuchs (Periphyton)
Segments of U. australis and U. macrorhiza were cultivated in glass petri dishes for 2—4 weeks. Periodical observations of the sessile and vagile animals staying on the plants led to an approximate quantitative stock-taking of the Aufwuchs for which the Utricularia itself is the substrate. The bladders of preserved specimens were also examined for adhering animals. Even in the fixative there could be found organisms which had been between the plants.
2.3. Treatment of plants before examination
As a reference to the fixated specimens, fresh material was examined, except U. vulgaris. Separated branches of the bladderworts were stored in petri dishes filled with water from their habitat. The traps were opened within 4 days after sampling. Additionally, Utricularia-segments were fixed in 4 % formalin.
2.4. Opening of the traps and determination of the contents
Living as well as fixated bladders were separated from the plant with a scalpel under the binocular magnifier and placed in some water on a slide. With two fine sharpened insect needles the bladder walls were carefully torn apart and rent to pieces or were ripped up. This method proved to be more protective for the contents than to cut open the bladders with the scalpel. The opened bladders were studied with the microscope.
The animals found were determined and counted; the determination resulted according to the condition of the different specimens. Not every prey item could be determined up to the level of species.
If parts of the bladder-contents could be classed unequivocally with a systematic group because of certain characteristics, they were counted accordingly in the evaluation. For lack of certainty, digestive rests of unknown origin were not counted as animals but were classified as “detritus”. Traps containing nothing but detritus were not counted as empty. This category was reserved for bladders in which there was (at a magnification of 10×12.5) in fact no contents to notice.
3. Origin and composition of the bladder-contents
3.1. Aufwuchs
The abundance of the organisms to find on and between the plants is named with the nomenclature usual in limnology: single individuum, few, few to medium, medium, medium to many, many, mass occurrence.
3.1.1. Utricularia vulgaris from the Stallberger Teiche (“Pond 1”)
- Arcella discoides (Testacea, Arcellidae), many
- Lecane pyriformis (Dad.) (Rotatoria, Lecanidae), medium to many
- Lepadella spec. (Rotatoria, Brachionidae) medium
- Floscularia pedunculata (Joliet) (Rotatoria, Flosculariidae), medium
- Lumbriculidae (Clitellata, Oligochaeta), few to medium
- Armiger crista (L.) [Syn. Gyraulus crista L.] (Gastropoda, Planorbidae), few to medium
- Stylaria lacustris (L.) (Clitellata, Oligochaeta), few
- Hydra viridis Pall. [Syn. Chlorohydra viridissima (Pall.)] (Hydrozoa, Athecata), single individuum
3.1.2. Utricularia vulgaris from the Stallberger Teiche (“Pond 2”)
- Arcella discoides (Testacea, Arcellidae), many
- Centropyxis marsupiformis (Testacea, Centropyxidae), medium to many
- Floscularia pedunculata (Joliet) (Rotatoria, Flosculariidae), medium
- Chironomidae (Insecta, Diptera), larvae, few
- Lumbriculidae (Clitellata, Oligochaeta), few
- Ephemeridae (Insecta, Ephemeroptera), larva, non det., single individuum
3.1.3. Utricularia australis from the fen biotope
- Chydorus sphaericus (O.F.Müll.) (Crustacea, Cladocera), mass occurrence
- Copepod nauplii (Crustacea, Copepoda), mass occurrence
- Cyclopidae (Crustacea, Copepoda), many
- Floscularia pedunculata (Joliet) (Rotatoria, Flosculariidae), many
- Lepadella spec. (Rotatoria, Brachionidae), medium to many
- Lecane pyriformis (Dad.) (Rotatoria, Lecanidae), medium to many
- Notodromas monacha (O.F.Müll.) (Crustacea, Ostracoda), few to medium
- Monommata longiseta (O.F.Müll.) (Rotatoria, Notommatidae), few
- Chironomidae (Diptera, Nematocera), larvae, few
- Ceratopogonidae (Insecta, Diptera), larvae, few
- Actinophrys spec. (Heliozoa, Actinophryidae), single individuum
- Squatinella rostrum (Schm.) (Rotatoria, Brachionidae), single individuum
- Hydrachna spec. (Acarina, Hydrachnellae), single individuum
3.1.4. Utricularia macrorhiza, Pond “Bienenkunde”
- Chydorus sphaericus (O.F.Müll.) (Crustacea, Cladocera), many
- Lepadella spec. (Rotatoria, Brachionidae), many
- Paramecium bursaria Ehr. (Ciliata, Holotricha), many
- Stentor polymorphus (O.F.Müll.) (Ciliata, Heterotricha), medium to many
- Vorticella microstoma Ehr. (Ciliata, Peritricha), medium to many
- Cephalodella sterea (Gosse) (Rotatoria, Brachionidae), many
- Cyclopidae (Crustacea, Copepoda), few to medium
- Turbellaria, non det., few
- Euplotes spec. (E. charon?) (Ciliata, Hypotricha), few
- Hypotricha (Urostyla or Keronopsis), few
- Aspidisca spec. (Ciliata, Hypotricha), few
- Daphnia longispina (O.F.Müll.) (Crustacea, Cladocera), single individuum
Several times, Cephalodella, Lepadella, and Stentor could be observed directly before or on an Utricularia's trap door; one Stentor sat on the trap door of a bladder with side walls compressed.
3.2. Plankton
The filtrate of 10 l contained the following animals (mean value from 2×10 litres):
3.2.1. Utricularia australis from the fen biotope
- > 2,000 copepod nauplii (Crustacea)
- > 700 Cyclopidae (Crustacea, Copepoda)
- 3 Chydorus sphaericus (O.F.Müll.) (Crustacea, Cladocera)
- 12 Ephemeridae (Insecta, Ephemeroptera), larvae
- 13 Chironomidae (Insecta, Diptera), larvae
- 2 Culicidae (Insecta, Diptera, Genus Culex or Aedes), larvae
- 1 Chaoboridae (Insecta, Diptera), larva
- 4 Ceriodaphnia spec. (Crustacea, Cladocera)
- 2 Piona spec. (Acarina, Hydrachnellae)
3.2.2. Utricularia macrorhiza, Pond “Bienenkunde”
- 36 copepod nauplii (Crustacea)
- 33 Cyclopidae (Crustacea, Copepoda)
- 24 Calanoida (Crustacea, Copepoda)
- 6 Daphnia longispina (O.F.Müll.) (Crustacea, Cladocera)
- 2 Alona rectangula Sars (Crustacea, Cladocera)
- 2 Bosmina spec. (Crustacea, Cladocera)
- 6 Cloeon spec. (Diptera, Ephemeroptera), larvae
3.3. Trap contents
The comparison between traps opened living and fixated yielded no differences, thus the results are summed up.
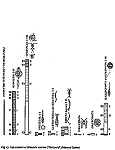
The distribution of the contents among the different organism groups is presented in the figures 1—5.
 |
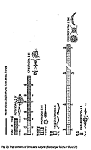 |
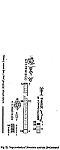 |
 |
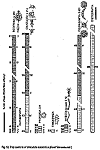 |
| Fig. 1 | Fig. 2 | Fig. 3 | Fig. 4 | Fig. 5 |
Explanation of Figs. 1-5:
Extra bold numerals (left) code the systematic groups as follows, light numerals (right) the numbers of individuals found.
- 1 non det.;
- 2 Bdelloidea, non det.;
- 3-11 Monogononta: 3 non det., 4-8 Brachionidae: 4 non det., 5 Keratella cochlearis, 6 Colurella spec., 7 Lepadella spec., 8 Mytilina spec., 9 Lecanidae: Lecane spec., 10 Notommatidae: Cephalodella spec., 11 Trichocercidae: Trichocerca spec.;
- 12-13 Copepoda: 12 Nauplius, 13 Cyclopidae, non det.,
- 14-19 Cladocera: 14 Chydoridae, non det., 15 Chydorus sphaericus, 16 Acroperus harpae, 17 Camptocerus recirostris, 18 Percantha truncata, 19 Alona rectangula
- 20 Ostracoda: Cyclocypris spec.
21-28 Testacea: 21 Arcella spec., 22 Arcella discoides, 23 Arcella vulgaris, 24 Centropyxis marsupiformis, 25 Centropyxis aculeata, 26 Euglyphidae, non det., 27 Trinema spec., 28 Difflugia corona;
29-30 Ciliata: 29 Litonotus spec., 30 Ophryoglena spec.;
31 Gastrotricha: Chaetonotus spec.; 32 Nematoda non det.; 33 Tardigrada: Heterotardigrada, non det.;
34-35 Chelicerata: Acarina: 34 non det., 35 Unionicola spec.;
36-40 Insecta (larvae): 36 Diptera, non det., 37 Ceratopogonidae, non det., 38 Chironomidae, non det., 39 Culicidae (genus Aedes or Culex); 40 Ephemeridae, non det.;
- 41-43 Chlorococcales: 41 Pediastrum duplex, 42 Pediastrum boryanum, 43 Scendesmus spec.;
44-46 Desmediales: 44 Closterium spec., 45 Cosmarium spec., 46 Cosmarium formosulum; - 47-48 Euglenophyta: 47 Euglenales: Phacus tortus, 48 Peranemetales: Heteronema spec.;
49 Diatomeae: Amphipleura spec.;
50-52 Dinophyta: 50 Peridinium spec., 51 Peridinium tabulatum, 52 Peridinium cinctum
4. Discussion
Due to the partly rapid digestion up to detritus, a lot of organisms could not longer be determined and were therefore not counted. As certain taxa are digested faster than others — e. g. ciliates and bdelloid rotifers can after a few hours no longer be taken into account, while crustaceans and armoured rotifers may still be identified after several days — they are not sufficiently represented in the evaluation. Therefore, the prey composition from the bladders conveys a partly distorted image.
Resulting from the advanced digestion status, a determination up to the level of species was not possible for a majority of the specimens found, the less so since some similar chidorids and brachionids occurred in coexistence. The prey animals seem to be decomposed in the bladders to a high degrade. A lot of particles, for lack of unequivocality classified as "detritus", seem clearly to have been animals before. Different grades of digestion of some cadavers which could be classified showed an almost indefinitely variable passage to undeterminable parts of detritus. In this context the supposition appears, that even the exoskeletons of arthropods might be digested completely when staying long enough in the bladder.
The comparison between the surrounding plankton and the Aufwuchs demonstrates that the bladderworts do not trap the animals swimming in the open-water but those organisms staying as vagile Aufwuchs between and on the plants. Cyclopoid copepodids are the one principal exception. They remain in free water but visit the periphyton for feeding and laying eggs.
The food spectrum of aquatic species consists mainly of these cyclopoid copepodids and copepod nauplii and a lot of monogonont Rotatoria, furthermore little cladocerans grazing bacteria, diatoms, and other algae on the surface of the plants.
Experiments of Seine, Porembski, Balduin, Wilbert et Barthlott (in prep.) show that ciliates are attracted by terrestrial Utricularia's bladders with chemical, water soluble substances. Results from Meyers & Strickler (1978 and 1979) that in particular Chydorus sphaericus grazes thoroughly the antennae and bristles of the bladders and by this way is led to the trap door, were confirmed by our own observations. Trap contents show, that indeed those grazing animals predominate. For aquatic bladderworts, an attraction based on behaviour seems to be a more important strategy than the engagement of chemical attractants. The much smaller monogonont rotifers behave in a similar way; as do chironomid larvae, which often even penetrate old, no longer functioning bladders.
Due to this catching practice, the capture of Culicidae larvae and Schistosoma mansoni miracidia and cercariae — in view of pest control considered by Brumpt and Langeron (both 1925), Angerilli & Beirne (1974 and 1980), Baumgartner (1987), and Gibson et Warren (1970), respectively — seems to be insignificant in normal biocenosae.
Protozoans could be found relatively seldom in the traps of the examined plants — most of them testaceans, only exceptionally ciliates.
As the results of U. australis from the fen biotope and the plant pond, and the two samples of U. vulgaris from the Stallberger Teiche indicate, bladderworts of the same species may prey upon different organisms in different habitats. This demonstrates, that aquatic Utricularia do not capture their food objects selectively but in dependence on the available prey community.
Lemmermann (1914), Schumacher (1960), Botta (1976), and others report on autotrophic organisms within the bladders. A lot of these organisms are not to be digested but to use the bladder as a habitat, in which they propagate and photosynthesise. Nothing of the kind could be ascertained at the examined specimens. On the contrary, it seems that different autotrophic organisms as well as metazoans and protozoans are part of the food spectrum, because they were regularly found in an advanced digested state.
Acknowledgements
The relevant authority, Oberkreisdirektor des Rhein-Sieg-Kreises, Amt für Natur und Landschaftsschutz, kindly granted an exceptional licence to sample the plants from the nature reserve Gagelbestand (Licence no. 67.1-32 95 45 01).
We thank R. Seine and S. Porembski for various support during work. We are grateful to A. Linkhorst-Brinkmann and I. B. Andrews for the critical review of the English version of the manuscript.
References
ANDRIKOVICS, S. / L. FORRÓ / E. ZSUNICS (1988): The zoogenic food composition of Utricularia vulgaris in the Lake Fertö.
– Opuscula Zoologica Budapest 23: 65—70
ANGERILLI, N. / B. BEIRNE (1974): Influences of some freshwater plants on the development and survival of mosquito larvae in British Columbia.
– Canadian Journal of Zoology 52: 813—815
ANGERILLI, N. / B. BEIRNE (1980): Influences of aquatic plants on colonization of artificial ponds by mosquitoes and their insect predators.
– The Canadian Entomologist 112: 793—796
BAUMGARTNER, D. L. (1987): Laboratory evaluation of the bladderwort plant Utricularia vulgaris (Lentibulariaceae) as a predator of late instar Culex pipiens and assessment of its biocontrol potential.
– Journal of the American Mosquito Control Association 3 (3): 504—507
BOTTA, S. M. (1976): Sobre las trampas y las víctimos o presas de algunas especies argentinas del género Utricularia.
– Darwiniana (Buenos Aires) 20: 127—154
BRUMPT, E. (1925): Capture des Larves de Culicidés par les Plantes du Genre Utricularia.
– Annales de Parasitologie Humaine 3 (4): 403—411
DARWIN, C. (1875): Insectivorous plants.
John Murray London
FRIDAY, L. E. (1989): Rapid turnover of traps in Utricularia vulgaris L.
– Oecologia 80: 272—277
GARBINI, A. (1899): Le vittime della Utricularia neglecta.
– Carlo Ferrari, Venezia
[cited after ANDRIKOVICS et al. (1988), HEGI (1918), MEISTER (1900)]
GIBSON, M. / K. S. WARREN (1970): Capture of Schistosoma mansoni miracidia and cercariae by carnivorous aquatic plants of the genus Utricularia.
– Bull. of the World Health Organization 42 (5): 833—835
HEGI, G. (1918): Illustrierte Flora von Mittel-Europa; mit besonderer Berücksichtigung von Deutschland, Oesterreich und der Schweiz.
VI. Band, 1. Hälfte
J. F. Lehmanns Verlag, München
JUNIPER, B. E. / R. J. ROBINS / D. M. JOEL (1989): The Carnivorous Plants.
Academic Press, London, San Diego
LANGERON, M. (1925): Biologie et Écologie des Utriculaires dans leurs Rapports avec la Prophylaxie du Paludisme.
– Annales de Parasitologie Humaine 3 (4): 412—425
LEMMERMANN, E. (1914): Algalogische Beiträge XIII: Über das Vorkommen von Algen in den Schläuchen von Utricularia.
– Abh. Naturw. Ver. Bremen 23: 261—267
MEISTER, F. (1900): Beiträge zur Kenntnis der europäischen Arten von Utricularia.
– Mémoires de l'Herbier Boissier 12: 1—40
MEYERS, D. G. / J. R. STRICKLER (1978): Morphological and behavioral interactions between a carnivorous aquatic plant (Utricularia vulgaris L.) and a chydorid cladoceran (Chydorus sphaericus O.F.M.).
— Verh. Internat. Verein. Limnol. 20: 2490—2495
MEYERS, D. G. / J. R. STRICKLER (1979): Capture Enhancement in a Carnivorous Aquatic Plant: Function of Antennae and Bristles in Utricularia vulgaris.
– Science 203: 1022—1025
SCHUMACHER, G. J. (1960): Further Notes on the Occurence of Desmids in Utricularia Bladders.
– Castanea 25: 62—65
TAYLOR, P. (1989): The genus Utricularia: a taxonomic monograph.
Kew Bulletin Additional Series XIV. Her Majesty's Stationery Office, London
WILBERT, N. (1969): Ökologische Untersuchung der Aufwuchs- und Planktonciliaten eines eutrophen Weihers.
– Arch. Hydrobiol./Suppl. XXXV 4: 411—518
![]()
© Nikolai Mette, Norbert Wilbert, Wilhelm Barthlott 2000, 2002